The Function of the Calciferous Glands of Earthworms
The calciferous glands, Kalkdrüsen, or glandes de Morren, are various terms applied to certain specialisations of the oesophageal epithelium which occur in most terrestrial oligochaetes. They are well developed in the family Lumbricidae.
In Lumbricus terrestris (Linn.) the calciferous glands are present in segments X-XIV, and consist of a pair of oesophageal pouches in segment X and a pair of oesophageal glands in each of segments XI and XII. In segment X the oesophageal epithelium is raised up into long narrow longitudinal folds projecting into the lumen. The tips of these folds fuse, enclosing a series of longitudinal tunnels elongated radially in transverse section. The tunnels extend backwards from the posterior part of segment X to segment XIII where they become smaller, and they end blindly in segment XIV.
The cells of the lamellae separating the tunnels are the secretory cells, and in the oesophageal glands they form a milky calcareous secretion, which passes forwards in the tunnels of the oesophageal wall to the pouches. There the individual particles coalesce and crystallise out as concretions of calcium carbonate, which escape into the oesophagus, pass down the gut, and finally reach the soil.
Since Julius Leo (1820) first described the glands many theories have been advanced as to their function.
Morren (1829) thought they might be connected with either the reproductive or the digestive system, while Lankester (1864) suggested that the oesophageal pouches “may be connected with the formation of the egg capsule” which at that time was considered to be calcareous. The milky secretion of the hinder glands he thought might be concerned in the process of digestion.
Claparède (1869) concluded that the function of the glands was the production of concretions which would help to triturate food in the gizzard. Both Perrier (1874) and Darwin (1881) agreed that this function must be a very subsidiary one, since small stones are already present in the earth which worms ingest. Darwin maintained that the calciferous glands “served primarily as organs of excretion”, but that the calcium carbonate would incidentally neutralise any humic acids arising by the decomposition of half-decayed leaves in the intestine.
Robinet (1883) and Harrington (1899) supported the neutralisation theory, but Harrington found no support for the excretionary view from his experiments. Com-bault (1909) regarded the glands as respiratory organs, absorbing oxygen and excreting carbon dioxide “fixed” as carbonate. M’Dowall (1926) also regards the fixation of carbon dioxide as the chief function of the glands.
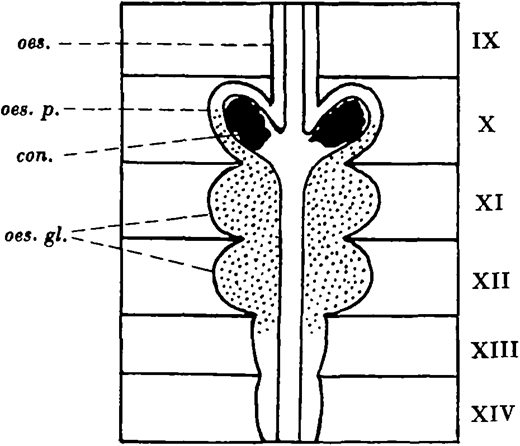
The calciferous glands of Lumbricus terrestris: con. concretion; oes. oesophagus; oes. gl. oesophageal glands; oes.p. oesophageal pouch.

The calciferous glands of Lumbricus terrestris: con. concretion; oes. oesophagus; oes. gl. oesophageal glands; oes.p. oesophageal pouch.
Michaelsen (1895, 1928), however, believes the primary function of the calciferous glands, or “Chylustaschen” as he calls them, is the absorption of food products. Gieschen (1930) gives the glands a respiratory significance. He believes they remove calcium bicarbonate which has accumulated in the blood as a result of the binding of carbon dioxide.
Voigt (1933) considers that the importance of the glands lies in the fact that when the carbon dioxide content of the environment is high the chalk can be used to eliminate that gas. That the buffering action of the secretion is important in the maintenance of reaction under such unfavourable conditions as a high carbon dioxide concentration in the surrounding medium or the presence of acid food is supported experimentally by Dotterweich (1933).
Since the experimental evidence on which the older hypotheses of Darwin and Michaelsen are based is both slight and contradictory, it seemed desirable to reexamine them and also the view that the glands have a respiratory significance.
II. CHEMICAL AND PHYSICAL NATURE OF SECRETION
The nature of the secretion of the calciferous glands in worms other than the Lumbricidae seems to be little known. In that family it consists of calcium carbonate. As Kelly (1901) pointed out, the carbonate in the two posterior oesophageal glands is in the amorphous state, while it is present in the anterior oesophageal pouches as crystalline concretions of calcite.
The calcareous spherules formed in the oesophageal glands have a diameter of 0·75–5μMost of the concretions are 0·5−1 mm. in diameter, but larger ones up to 2 mm. are not uncommon. Microchemical tests for calcium, magnesium, barium, strontium, ammonium salts, nitrates, phosphates and oxalates were applied to the amorphous secretion of specimens of Lumbricus terrestris, Allolobophora caliginosa, and Eisenia foetida. The secretion in all cases consisted of calcium carbonate, the tests for other radicles being negative except that a slight phosphate reaction was obtained in each case. No trace of ammonium salts was found with Nessler’s reagent even in specimens of Eisenia freshly collected from manure, this being contrary to Combault’s identification of ammonium carbonate in this worm. His test consisted in the recognition and distinction of crystals of calcium and ammonium chloroplatinates and must be regarded as of doubtful validity.
The crystalline concretions formed by Lumbricus terrestris consist wholly of calcium carbonate and a little organic matter. In section, viewed under a petrological microscope the crystals were recognised as calcite by the optical properties of high double refraction and twinkling, and distinguished from aragonite by the presence of the black cross under polarised light indicating a uniaxial crystal. The crystals differ from the concentric calcospherites described by Keilin (1921) in insect larvae by their irregular concretionary form, and in the presence of organic matter acting as a nucleus and scattered throughout the crystal. A few of the crystals obtained were, however, almost perfect rhombohedra.
Several analyses of the percentage of carbonate in the concretions were made by estimating their carbon dioxide content gravimetrically. It was found to be 94·9−96·7 per cent.
There is the possibility that some of the amorphous secretion may escape into the oesophagus as well as the concretions, and in two cases the amount was determined. Several earthworms were fed on moistened calcium sulphate which they ingested as they would earth. At the end of a fortnight the concretions greater than 0·33 mm. were sieved from the casts and analysed for carbonate. The carbonate still present in the casts was also determined.
In these two cases the amount of secretion other than the concretions is small.
The physical nature of the excreted carbonate
III. FEEDING EXPERIMENTS
The purpose of the following experiments was to ascertain (1) whether the calciferous glands of earthworms fed on various calcium salts continued to secrete calcium carbonate, and (2) whether the glands secreted carbonate when leaves were the sole source of calcium.
In the first experiments individual worms which had been kept 3 days in moistened filter paper were fed on moistened calcium salts, decaying pear leaves, 1 moorland peat (pH 4·2), and filter paper. At intervals equal portions of pear leaf were given to the worms as food. The carbonate concretions were collected, usually at the end of successive 5-day periods, by putting the casts through a sieve which retained particles of 0·5 mm. in diameter and over. After a considerable time the calciferous glands of the worms were fixed and sectioned, and the concretions present in the intestine counted.
The results given in Table II show that the glands continue secreting when worms are kept feeding on the carbonate or the phosphate of calcium, and also on pear leaves, since concretions continued to be formed and passed out. The worm in filter paper ceased forming concretions within a week, but at the end of 14 days calcareous spherules were still present in a section through the oesophageal glands. The worm kept in calcium oxalate ingested very little of it and seemed to be affected by the poisonous nature of the salt. The acid peat seems to have acted directly on the glands of the worm kept in it, since few calcareous spherules were present and fragments of protoplasm were found in the lumen of the glands, apparently owing to the disruption of the gland cells.


The concretions in each case consisted of calcium carbonate. The numbers of concretions found have no special significance, since the concretions varied in size. It is probable that the calcium carbonate secreted during the first few days of the experiments was already present in the worms.
In a second series of experiments an approximate measure of the carbonate excreted was obtained by weighing the concretions formed by two worms in periods of a week. Tap water was used to moisten the salts, but the amount of calcium present in it could not account for more than about 1 mg. of carbonate weekly.

Experiment CaSO4 (1) was carried on for 8 weeks, the worms remaining healthy and continuing to form concretions.
It seems clear from the preceding experiments that the inorganic carbonate, sulphate, and phosphate are available as a source of calcium for the secretion of the glands. In leaves both inorganic and organic calcium compounds are present, but whether only the former or both are available to worms has not been shown. Feeding experiments with several organic calcium compounds were unsuccessful.
IV. X-RAY PHOTOGRAPHY
Voigt (1933) has used X-rays for studying the calciferous glands. The following observations and experiments were almost completed before the writer’s notice was drawn to his paper.
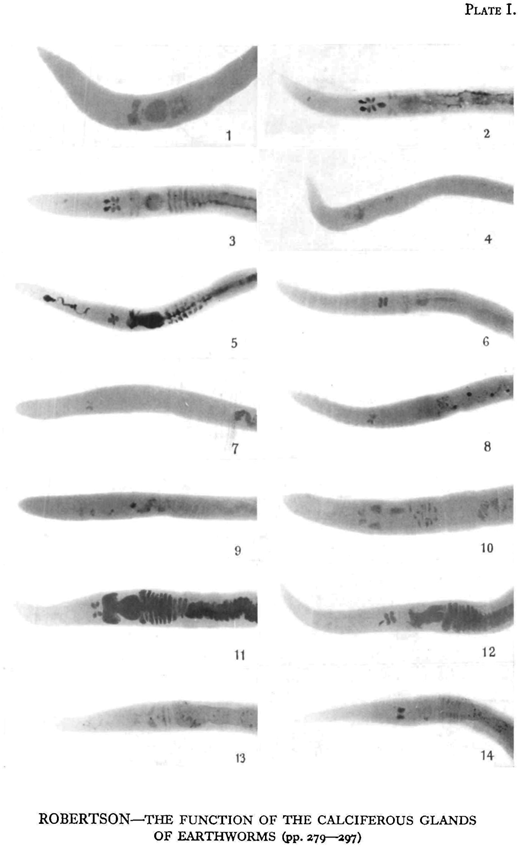
The physiological state of the glands in earthworms kept in various media can be determined at any time by X-rays. When in a state of secretion the small calcareous particles present in the glands cause them to stand out when the worm is X-rayed. The more calcium present in the glands and the more compact it is, the more opaque are they to the rays.
The glands of specimens of Lumbricus terrestris collected from garden soil, grass lawns, and from pasture over chalk were all in a state of active secretion.
Attempts were made to eliminate most of the calcium from the glands by keeping earthworms in such acid media as filter paper moistened with lemon juice, starch, acid potassium phosphate, and peat. Keeping them in acid peat was the most successful, as after about 10 days the glands were practically free from calcium and no longer stood out in the X-ray plate. At this stage the earthworms were X-rayed, and then put into various calcium salts moistened with distilled water. Controls were kept feeding on filter paper, starch, and peat. Plates were taken at intervals of 2-5 days.
The X-ray technique was as follows : 30 kV., 140 mA., 1 1 0 sec. at 12 in. distance, using a fine-focus Coolidge Tube supplied by a four-valve transformer unit.
As shown in Plates I and II, the glands of the worms used were practically free from calcium after 11 or 13 days in peat. Two days afterwards, the glands of the worms kept in the carbonate, sulphate, phosphate, and chloride of calcium were markedly denser, showing that secretion had taken place, while in those worms kept as controls in filter paper, starch, and peat the density of the glands had only slightly increased. Concretions had already been formed in the glands of the worm fed on calcium carbonate. After 4 or 5 days concretions were present in the oesophageal’ pouches of the worms fed on calcium chloride and on calcium sulphate.
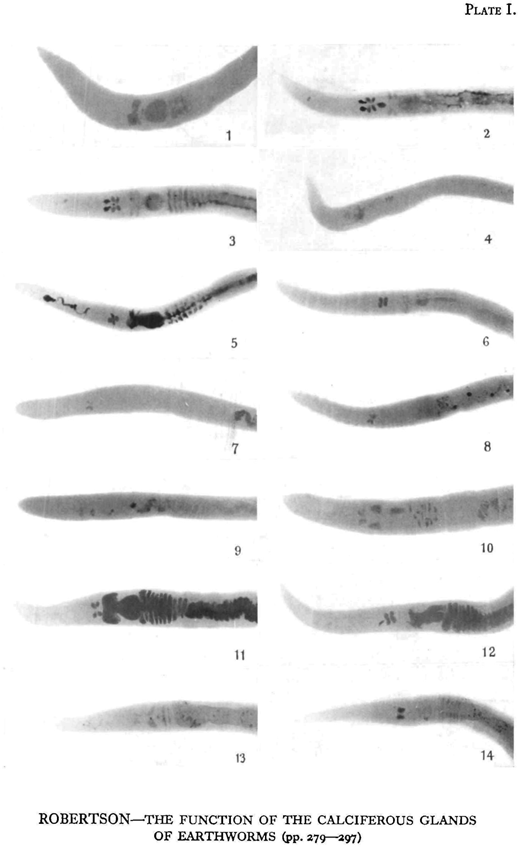
ROBERTSON—THE FUNCTION OF THE CALCIFEROUS GLANDS OF EARTHWORMS (pp. 279—297)

ROBERTSON—THE FUNCTION OF THE CALCIFEROUS GLANDS OF EARTHWORMS (pp. 279—297)
The worm in calcium oxalate also showed secretion, this result being contradictory to that obtained in the feeding experiments. It seems that the result obtained there must be disregarded, since the section through the glands was made after the worm had been 14 days in the salt and was apparently affected by its poisonous nature.
The experiments as a whole indicate that some absorption of the ingested salts takes place, resulting in an increased concentration of calcium in the blood. This results in an active excretion of the excess calcium as carbonate by the calciferous glands.
V. HYDROGEN-ION CONCENTRATION OF GUT, SOIL, AND CASTINGS
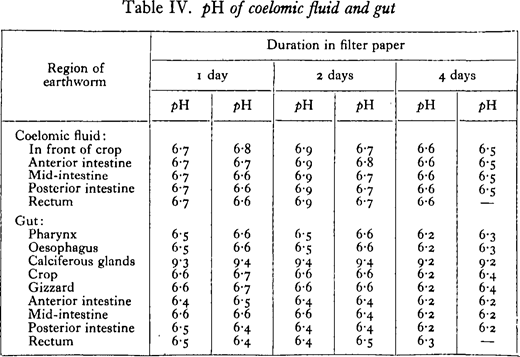
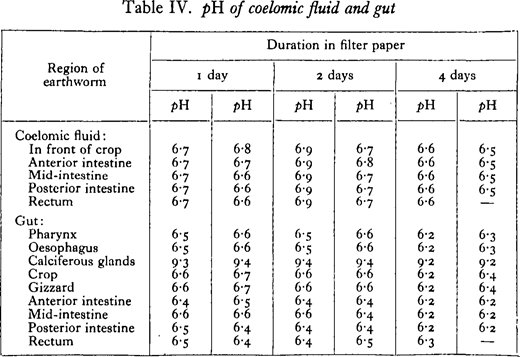
Since there seems to be no precise information about the reaction of the wall of the alimentary canal, the pH of the gut and of the coelomic fluid was determined by a colorimetric method.
Earthworms were kept in moistened filter paper to free them from ingested earth. After periods varying from 1 to 4 days, specimens were anaesthetised in chloroform vapour and dissected under liquid paraffin. An indicator was drawn into a capillary tube of about 1· 5-mm. bore. At a particular region of the coelom or gut it was expelled and drawn up again several times, this operation being conducted under the paraffin. The colour was finally compared with standards made up in similar tubes with buffer solution of known pH. The buffer solutions of Clark and Lubs were used, and also their indicators brom thymol blue, brom cresol purple, phenol red, and thymol blue.
From Table IV it is seen that there is not much variation in the pH of the different regions of the gut, most of the values lying between pH 6·3 and 6·6. A conspicuous exception is seen in the calciferous glands or rather the two hind oesophageal glands, where the ppH of the isolated secretion is usually about 9· 4. The reaction of the coelomic fluid is generally more alkaline than the gut wall. The table given is for specimens of Lumbricus terrestris, but results with Allolobophora caliginosa and Eisenia foetida were closely similar.

Salisbury (1924) has shown that worm casts are generally less acid than the soil from which they are derived. This holds only for acid soils, since he found a reduction in the pH of casts from worms living in alkaline soils. The percentage of carbonate in the casts was approximately double that of the soil, and he suggested that the calcareous particles from the calciferous glands neutralise the soil acids as Darwin had believed.
Salisbury’s data on the pH of worm casts were confirmed by keeping specimens of Lumbricus terrestris in well-mixed and sifted natural soils of different reaction. The pH of the casts and the soil was estimated each day by making an aqueous extract of soil and water in the proportion 1:5, adding indicator, and centrifuging the extract. The colour was then compared with that of standard solutions.
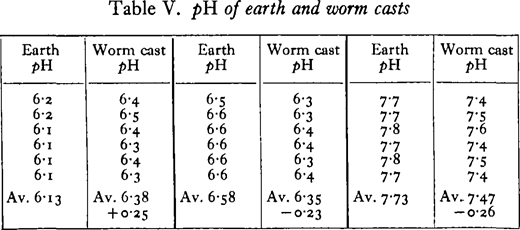

The difference in pH of the soil and cast refer to the moist soil and cast. When these were air-dried before the determinations, the difference was usually reduced in the case of the acid soil, and it disappeared entirely in the chalky alkaline soil.
Darwin had found that the intestinal wall of worms was acid to litmus, and also fresh casts except those from chalky soils. Casts i day old were usually no longer acid, and he attributed this to the breakdown of humic acids formed in the gut by the decomposition of organic material.
It is clear that the fall in the reaction of the soils of pH 7· 7 and 6· 6 is not due to the amorphous secretion (pH 9-4) of the calciferous glands. Concretions from the glands were present in the casts, but they do not affect the reaction in the slightest. From the data obtained on the reaction of the gut, it is suggested that the tendency towards neutralisation of the worm cast is due, mainly at least, to the secretions of the gut wall, the secretion of the calciferous glands having no appreciable effect owing to the form in which most of the calcium carbonate is passed out of the glands. In connection with the last point, it seems that the earthworm has no control over the form in which the carbonate is passed into the gut, since concretions of no value in neutralisation are formed whether it is kept in alkaline soil, acid soil, or feeding on decaying leaves. Powdered calcium carbonate immediately neutralises humus as Robinet observed, but concretions kept in acid peat (pH about 4· 2) did not alter its acidity and seemed to be quite unaffected by the humic acids even after a weeks
VI. OPTIMUM HYDROGEN-ION CONCENTRATION OF ENZYMES
Darwin, Robinet, and Harrington have suggested that the secretion of the calciferous glands will neutralise any acids present in the soil ingested or formed by the decomposition of organic matter in the intestine, and thus bring the contents of the gut to a more favourable reaction for digestion. The evidence that neutralisation is advantageous to digestion rests on the observations of Willem and Minne (1899), who found an extract of the intestine to digest fibrin most actively in an alkaline medium, while Lesser and Taschenberg (1908) found the proteolytic enzyme to be active in both weak acid and weak alkaline media.
It was thought of interest to investigate the optimum pH’s of the main intestinal enzymes. A crude distilled water extract of the intestine was used in the following experiments. Its pH was about 6·2–6·4
The sugars produced in amylase digestion were estimated by the Hagedorn and Jensen method as modified by Boyland (1928). Fatty acids were directly titrated with sodium hydroxide in a carbon-dioxide-free atmosphere with phenolphthalein methylene blue as indicator. When the buffer was suitable the acidimetric titration of Glick (1934) to a pH of 6· 6 with brom thymol blue was used. Amino-N was determined by the formol titration, the formaldehyde being added after neutralisation to phenolphthalein.
The duration of the amylase experiment was 23 hours, the average time taken for filter paper to pass through the gut in a number of worms at room temperature.
Amylase digestion
Substrate, 2 per cent, starch solution, 5 c.c. ; enzyme extract, 2 per cent., 5 c.c.; buffer solution, M c Ilvaine’s, 5 c.c.; toluene; 23 hours at 17· 5° C.; controls, boiled extract; pH’s measured at beginning and end of experiment with B.D.H. Capillator, no change after incubation; figures for experiments subtracted from average of controls.
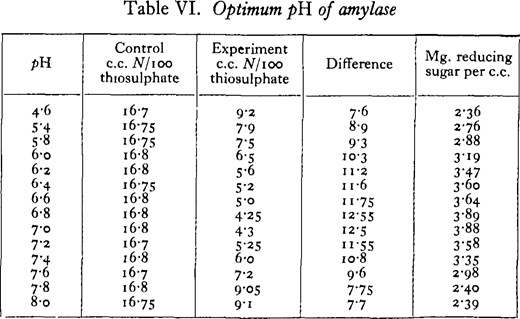

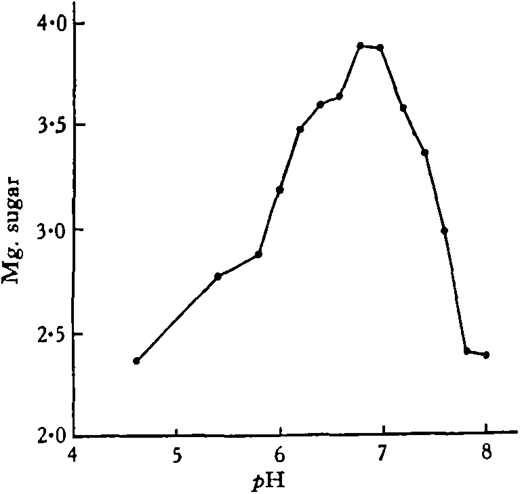

Under the conditions of the experiment, the optimum reaction for amylase digestion lies between pH 6· 8 and 7· 0.
Since the calcium carbonate secreted by the glands may increase the activity of the amylase, the following experiment was performed.
Substrate, enzyme extract and buffer solution as before; CaCO3, powdered (p) or as concretions (c); 23 hours at 17-18° C.;pH 6· 8.
Calcium carbonate seems to have no specific effect on amylase digestion either in powdered form or as concretions. Initially when the powdered carbonate was added, it caused a change in the pH of the buffer solution owing to its alkalinity. Thus 5 c.c. of buffer solution of approximately pH 6-6 were added to the substrate, and the addition of the powdered carbonate brought the pH to 6-8. The concretions did not affect the pH.
Calcium carbonate and amylase digestion
Lipase digestion
10 c.c. Clark’s phosphate-NaOH buffer; i c.c. 2 per cent, enzyme extract; 0· 5 c.c. methyl butyrate; 9 hours at 18 0 C.; enzyme extract added to controls immediately before titration with phenolphthalein methylene blue as indicator.
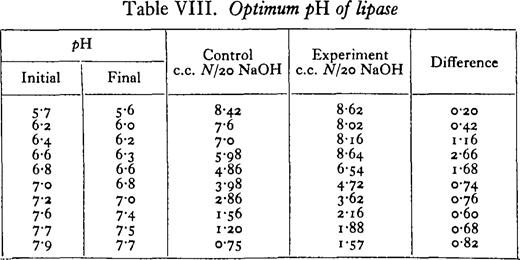

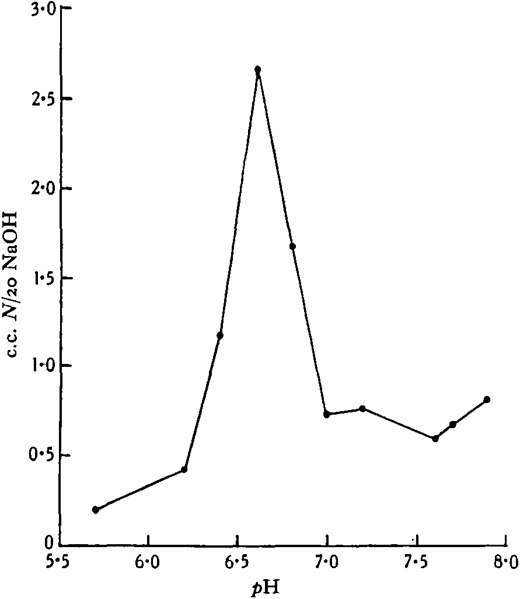

The above is one of several experiments in which the optimum pH in phosphate buffer varied from pH 6-4 to 6-6 with different extracts. When a borate buffer was used, an increase in the liberation of fatty acids up to the alkaline limit of the buffer was observed. With ethyl butyrate as substrate, optima exactly similar to those obtained with methyl butyrate were found, but with the neutral fat tributyrin a more alkaline optimum was found in the phosphate buffer, pH 7· 3-7 – 7.
Proteinase digestion
Unfortunately the proteinase even in a 20 per cent, extract of the intestine was very weak, and sufficient digestion at 18 0 C. in 24 hours could not be obtained in order to determine any optima. However, when the digestion of casein or gelatin in Clark’s phosphate buffer was prolonged at 25 0 C. for 48 hours, there was more digestion at a pH of about 8-o than at either pH 6· o or 7· 0.
It is realised that the optimum pH’s of these enzymes may vary under different conditions. Nevertheless, 50 per cent, of the activity of the enzymes has taken place between pH 6· o and 7· 5 in the case of the amylase, and between pH 6· 4 and 6· 9 in the case of the esterase. Only in the case of the weak proteolytic enzyme is an alkaline medium apparently specially favourable to digestion.
The reaction at which digestion occurs in the earthworm will largely depend on the amount of soil present in the gut and its acidity or alkalinity. Decomposing organic matter in the gut and the products of digestion may also be expected to influence the reaction in some cases. Salisbury has found earthworms in soils ranging from pH 5· 1 to 7· 5, while Allee and co-workers (1930) have found them in soils varying from pH 5· 6 to 8· 3. They are most abundant in soils of approximately neutral reaction. That the reaction of the soil can be slightly altered in the gut is shown by the tendency towards neutralisation shown by worm casts, but this would appear to be of only small benefit in digestion. But the secretion of the calciferous glands certainly plays no dominant part in the neutralisation, and the suggestion of Darwin and Harrington concerning this cannot be substantiated.
VII. AMOUNT OF CARBON DIOXIDE BOUND AS CARBONATE
It has been shown that the calciferous glands of earthworms which have ingested various calcium salts secrete calcium carbonate, which passes into the gut as calcite concretions. Since in all cases, except the carbonate, the salt of calcium seems to be changed in its intermediary metabolism to carbonate, it is inferred that the carbonate radicle has come ultimately from the carbonic acid of the blood. The amount of carbon dioxide bound in this way as carbonate can be measured by estimating the carbonate present in the worm casts, and can be compared with the respiratory carbon dioxide.
In each of these experiments two worms were used as a unit. They were kept in filter paper moistened with 5 c.c. of a dilute solution of calcium nitrate or chloride and 2 c.c. of a nutrient solution. 1 Others were kept in the sulphate and carbonate moistened with distilled water and 2 c.c. of the nutrient solution, or feeding on pear leaves. The worms were kept for over a week in their respective salts before any measurements were made. The experiments were conducted in a constant temperature room at i8° C., the worms being in darkness the whole time.
On three alternate days the respiratory carbon dioxide of each pair of worms was measured in a Haldane type of respiration apparatus. Carbon-dioxide-free air saturated with moisture was passed over the vessel containing the worms, dried in calcium chloride U-tubes, and the respired carbon dioxide absorbed in soda-lime U-tubes. Blank experiments were carried out between the respiratory measurements, the correction being o· 2 mg. or less. At the end of the week the casts were analysed for carbonate in the same apparatus, by adding hydrochloric acid and expelling the carbon dioxide by gentle heating while carbon-dioxide-free air was passed through. The apparatus was again checked by analysing small amounts of pure calcium carbonate of similar order to that found in the casts, the results being correct within 2 per cent. In most cases the whole experiment was repeated with the same worms. In the casts of the worms in calcium carbonate the concretions only could be analysed.
Since the factor of movement was uncontrolled there was naturally considerable variation in the amounts of carbon dioxide respired, the mean deviations from the figures given in the table ranging from o-8 to io-o per cent, with an average of 5· 1 per cent.
Amount of carbon dioxide bound as carbonate
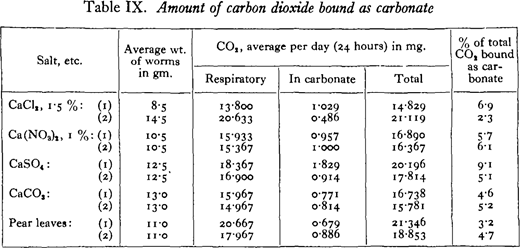

As might have been expected the results vary considerably even in the case of duplicate experiments with the same worms. But in no case has the amount of carbon dioxide excreted as carbonate exceeded 10 per cent, of the total, the average amount being 5· 3 per cent.
Under natural conditions it is probable that this percentage will vary widely at different times. The carbonate excretion will depend on the presence of suitable calcium compounds in the earth and organic matter ingested, and the various factors influencing their absorption and excretion, while the amount of carbon dioxide respired will vary with the muscular activity of the worms, with changes in the temperature and humidity of the environment, and also with the degree of starvation.
It would seem improbable, however, that the excretion of carbon dioxide bound as carbonate could ever account for more than a fraction of the total metabolic carbon dioxide.
VIII. THE SUPPOSED ABSORPTIVE FUNCTION
Michaelsen has repeatedly stated that the primary function of the calciferous glands or “Chylustaschen” is probably the absorption of food products.
Darwin demonstrated the presence of an amylase in the pharyngeal secretion, while Willem and Minne found a proteinase in an extract of the pharynx. It is also well known that some external digestion can take place in earthworms, leaves being often digested in this manner until only the network of veins remains.
Liebmann (1927) in investigating fat absorption found that only isolated cells in the calciferous glands absorbed the fat, absorption taking place chiefly in the intestine but also in the crop.
Attempts were made to demonstrate absorption by the iron saccharate technique used by Yonge (1926) and others, but they were not very satisfactory. A dilute solution of the salt was injected into the alimentary canal by way of the mouth, since filter paper moistened in it was not ingested at all.
When absorption did take place, groups of adjacent cells in the intestine were coloured blue by the Prussian blue reaction, but only a few scattered cells in the lamellae of the calciferous glands.
The great vascularity of the glands and the large surface afforded by the epithelial folding are advantageous to secretion and do not necessarily imply an absorptive function.
Stephenson (1930) remarks on the well-marked tendency in the elaboration of the glands for segregation from the alimentary canal culminating in the Lumbricidae, where connection is retained only by one end of a long tunnel, such a condition being obviously unsuitable for an absorptive function.
Michaelsen’s view can be upheld neither from the experimental evidence nor from physiological probabilities.
IX. DISCUSSION
In Darwin’s important work—Vegetable Mould and Earthworms—he argued that worms which consume fallen leaves containing usually a large percentage of lime would be liable to become charged with too much calcium were it not for its excretion as the carbonate by the calciferous glands. He observed that the glands of worms from mould over chalk “contained as many free calciferous cells (amorphous secretion), and fully as many and large concretions as did the glands of worms which lived where there was little or no lime ; and this indicates that the lime is an excretion and not a secretion poured into the alimentary canal for some special purpose”.
Harrington kept worms in calcium carbonate crystallised in minute rhomboids (to distinguish it from the amorphous secretion) and sectioned the glands after 2-5 days. Only a few particles were forming in the glands and no concretions were found in the intestine. He concluded that his experiments did not support the ex-cretionary view, but no great weight can be attached to them because of their short duration.
Combault states that he confirmed Harrington’s experiment with the carbonate but he gives no details.
Voigt found some earthworms in which the glands did not stand out when X-rayed, and he kept them in the carbonate, sulphate, and oxalate of calcium for 8-10 days. The glands did not appear even then in an X-ray plate, and he concluded that worms could not make use of these salts and that perhaps only organic calcium compounds in their food were available as a source of calcium for the secretion.
Voigt’s results are inconsistent with the experiments of the writer in which by various methods it has been shown that excretion of calcium carbonate is continued for considerable periods (from 14 days to 8 weeks) in the carbonate, sulphate, phosphate (tribasic), oxalate, chloride, and nitrate of calcium, thus affording evidence of the availability of these inorganic salts as a source of the calcium of the secretion. It is possible that organic compounds can be utilised also.
Emphasis has been laid by Combault and M’Dowall on the fact that carbon dioxide is being excreted bound as carbonate, and they maintain that it is this particular function that is important. Combault found an increased carbonate content of worm casts as compared with a sample of the original soil, and this has been fully confirmed by Salisbury. Combault further states that ammonium carbonate is found in the glands of Eisenia foetida of manure heaps (which the writer has failed to confirm), and that therefore the carbon dioxide is fixed by the most abundant base in the environment—lime in the soil and ammonia in manure. He believed that thé carbon dioxide thus fixed would otherwise be liable to asphyxiate the worms, but it is highly improbable that the relatively small amount which seems to be bound as carbonate has any importance in this connection. He also believed that the calciferous glands were the actual site of this fixation and that they constituted “un appareil respiratoire” absorbing oxygen as well. His special fixing and staining method to demonstrate oxygenation in the blood of the efferent vessels of the calciferous glands is questioned by Winterstem (1921). The assertion that water is drawn into a diverticulum of the oesophagus in front of the glands, and passed backwards between the lamellae to re-enter the oesophagus by a second opening in segment XIV is inaccurate. This opening has never been found by subsequent workers including the writer, and there is never sufficient water present in the oesophagus to justify even the nominal correctness of the term “branchies internes” which he also gives to the calciferous glands.
Although the secretion would seem to represent a certain amount of bound carbon dioxide, the calciferous glands themselves are in no true sense respiratory organs at all.
In a recent attempt to demonstrate a connection of the calciferous glands with the respiratory function Gieschen measured by means of a Krogh respiration manometer the respiratory quotients of worms kept under different conditions.
When worms were fed on chalk the R.Q. was lowered. Gieschen interprets this as indicating that chalk absorbed into the blood has combined with carbon dioxide according to the reaction
but the possibility that the reaction has taken place not in the blood but in the gut between respired carbon dioxide and ingested chalk must not be disregarded.
Gieschen also found that the R.Q. was lowered when worms were kept in atmospheres of high carbon dioxide content, and that in experiments of 3 days’ duration the ’R.Q. after falling became stationary when the percentage of carbon dioxide reached 20 per cent, or higher. Since the R.Q. in similar experiments of worms in which the glands had been removed increased to nearly unity, due apparently to a sinking of oxygen consumption, he concluded that the glands made oxygen absorption possible in high carbon dioxide tensions, and prevented the tension of the latter gas in the blood from exceeding a certain limit. The glands are supposed to achieve this latter function by removing the bicarbonate which accumulates in the blood.
Gieschen’s work has been adversely criticised by both Voigt and Dotterweich, who point out that the basis of his reasoning is false, since the calciferous glands excrete the carbonate and not the bicarbonate of calcium. The apparatus he used is so sensitive that very small changes in the experimental conditions give rise to considerable differences in the measurements. It is also probable that at the high carbon dioxide tensions Gieschen used some of the respiratory carbon dioxide was retained in combination within the tissues of the earthworms, thus making difficult the interpretation of his respiratory quotients.
Voigt’s experiments on the availability of the carbonate, sulphate, and oxalate as a source of the calcium of the secretion have already been mentioned. By X-ray photography he confirmed the mode of excretion, the concretions in the anterior oesophageal pouches being formed from the amorphous carbonate in the oesophageal glands, and then passed into the oesophagus. He also found that the density of the oesophageal glands was much reduced when worms had been kept in an atmosphere containing 25 per cent, carbon dioxide for 3 days, or in one of 14 per cent, for 5 days, indicating that the carbonate had gone into solution in the blood as the bicarbonate. He asserts, however, that this bicarbonate escapes through the skin, a statement unsupported by any conclusive evidence.
In Voigt’s view, the biological importance of the glands rests on the fact that under unfavourable carbon dioxide conditions (over 14 per cent.) the chalk formed in the glands can be utilised in the elimination of that gas.
Dotterweich has brought forward considerable evidence to support his view that the calciferous glands act as a buffer reserve in the maintenance of reaction. Thus the pH of the coelomic fluid of worms in which the glands have been removed is lower than that of normal worms after being 3 days in an atmosphere containing 5 per cent, carbon dioxide. The reaction of the coelomic fluid of normal worms fell only slightly after 16 hours in 25 per cent, carbon dioxide. Analyses of the calcium content of worms, excluding gut and glands which were dissected out before each analysis, showed an increased amount per unit weight after they had been kept in high carbon dioxide percentages. This seems to indicate that the calcium in the glands has been distributed throughout the body.
Dotterweich concludes that under unfavourable conditions such as a high percentage of carbon dioxide in the atmosphere or the presence of acid food in the gut, the buffering action of the glands comes into play, but under normal conditions the function of the glands is the storage and excretion of superfluous calcium salts.
The validity of the conclusions of these two authors depends on whether earthworms are ever exposed for long periods to such high percentages of carbon dioxide as were used in their experiments. The percentage of carbon dioxide in the’soil atmosphere depends largely on the activities of micro-organisms, and has an average value at the depth of 6 in. of 0-25 per cent. (Russel and Appleyard, 1915). During wet conditions, these authors found the high value of 9-1 per cent, in grassland under Festuca ovina. To this will be added the amount of carbon dioxide respired by the worm, but the aeration afforded by the burrow itself would in the writer’s opinion prevent any considerable increase of long duration in the carbon dioxide content of the burrow from that source.
The possibility that the calcium carbonate present in the calciferous glands might act as a buffer reserve under exceptional conditions must, however, be admitted.
The evidence adduced to support the neutralisation hypothesis is as follows :
- Histological appearances of “over-secretion” were found by Harrington after sectioning the glands of worms kept in acid media such as lemon juice, starch, sawdust, and calcium phosphate.
- Salisbury found that worm casts were generally more neutral than the soil from which they were derived.
- The proteolytic enzyme seems to be most efficient in an alkaline medium.
- The position of the glands suggests that they may have some function in digestion.
- Robinet pointed out that calcium carbonate neutralises humus.
“Over-secretion”—the cytoplasm of the gland lamellae reduced in thickness, crystals and shreds of protoplasm present in the gland cavities—is found also in the glands of worms kept in acid moorland peat, but the writer agrees with Combault in attributing this appearance to the direct action of the acids on the secretory cells, disrupting them, and not to an attempt on the part of the worm to neutralise the acids by increasing the secretion.
A partial neutralisation of acid soil does take place in the worm, but it would seem to be effected chiefly by the secretions of the gut other than that of the calciferous glands. The reduction in the pH of worm casts in alkaline and slightly acid soil demonstrating that the amorphous secretion is not being passed into the oesophagus, the analyses of the amount of carbonate excreted as concretions of no value in neutralisation, and the fact that concretions are still passed out when worms are kept in acid soil, peat, and decaying leaves, all point to the ineffectiveness of the secretion as an agent for neutralisation.
Only the weak proteolytic enzyme seems to be more efficient in an alkaline medium. The optimum hydrogen-ion concentrations of the stronger and more important amylase and lipase give no definite support to the hypothesis.
The position of the calciferous glands in the oesophagus suggests that the secretion may be of importance in intestinal digestion, and if the organs are solely for excretion it seems peculiar that their position is not in a more posterior part of the alimentary tract. However, the position is advantageous in that a considerable amount of blood from the dorsal vessel which has collected absorbed food products and salts from the dorso-intestinal vessels, courses directly through the glands.
The formation of carbonate concretions has the physiological advantage that the calcium is now in a form which is not reabsorbed by the intestine but passes unchanged to the exterior. It also means that no neutralisation can be effected, since the compact crystals are chemically inactive in such acidities as are likely to be experienced by earthworms.
The theory that the glands absorb food products has already been discussed, and it would seem that such a function is improbable, in the Lumbricidae at least.
X. CONCLUSIONS
There seems to be no adjustment of the amount of absorption of calcium salts to the needs of the earthworm as a whole, and when ordinary requirements for that element have been met, the excess is concentrated as carbonate in the calciferous glands and excreted in a relatively inactive form.
The formation of calcium carbonate is more or less continuous and takes place under acid, neutral, and alkaline conditions provided that calcium compounds are available in the soil and food ingested.
The carbonate radicle may often represent fixed metabolic carbon dioxide, and its elimination in conjunction with calcium by the calciferous glands points to the importance of these organs in connection with the acid-base balance of the blood.
Under conditions of a high, sustained carbon dioxide content in the environment, the carbonate in the glands will go into solution as the bicarbonate, thus acting as a buffer reserve in the maintenance of reaction.
There is no evidence that neutralisation of acid substances in the gut is a vital function or even an incidental process, and the theory of food absorption is quite improbable.
ACKNOWLEDGEMENTS
I wish to express my sincere thanks to Prof. Graham Kerr for suggesting this work and for his continued interest in it, and especially to Dr James Gray for advice and many helpful suggestions. My thanks are also due to Drs A. E. Barclay and D. W, Lindsay of the Radiology Department, University of Cambridge, for their co-operation and assistance in taking the X-ray plates.